What Is Organic Chemistry?
Types of hydrocarbons
What hydrocarbons do we study?
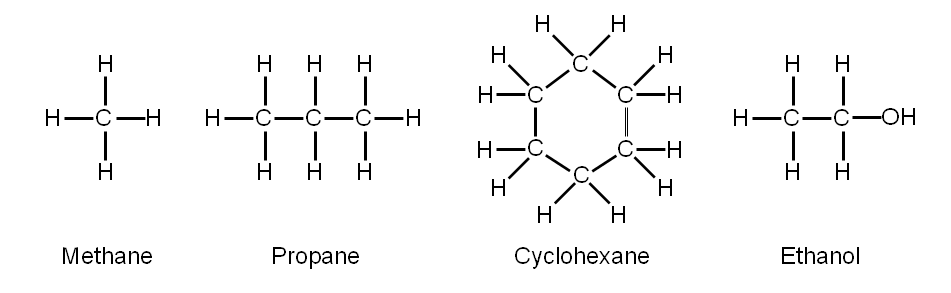
Hydrocarbons are organic compounds composed of carbon and hydrogen atoms. The hydrocarbons we study are arranged in straight chains, branched or non-aromatic ring structures structures.
These hydrocarbons are called Aliphatic. They do not have a pleasant odor. (You don’t need to know this).
What are Aromatic hydrocarbons?
Aromatic hydrocarbons are NOT in the Physical Sciences 3 course, but are described here for the sake of completeness.
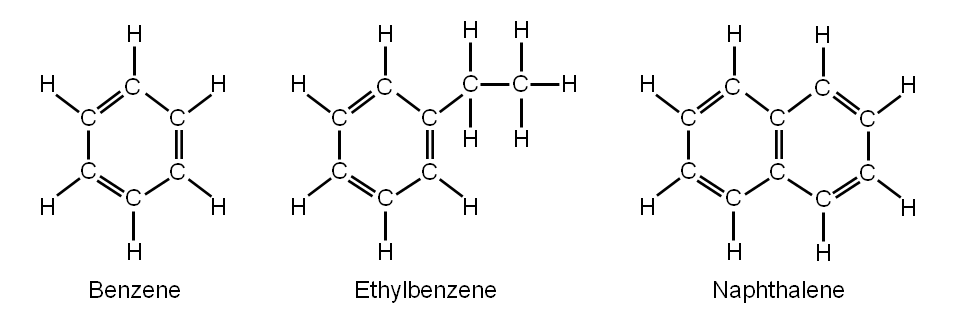
Aromatic hydrocarbons are organic compounds composed of carbon and hydrogen atoms, arranged in ring structures. They exhibit bonding that must be described using the resonance hybrid concept of valence bond theory or the delocalization concept of molecular orbital theory.
They have a pleasant odor, hence the name.
Introduction to alkanes
What are alkanes?
Alkanes are members of a homologous series.
(A homologous series is a collection of compounds with the same general formula that differ only in the carbon chain length.)
Alkanes are hydrocarbons with only SINGLE bonds between carbons.
The simplest alkanes have their C atoms bonded in a straight chain; these are called normal alkanes. They are named according to the number of C atoms in the chain.
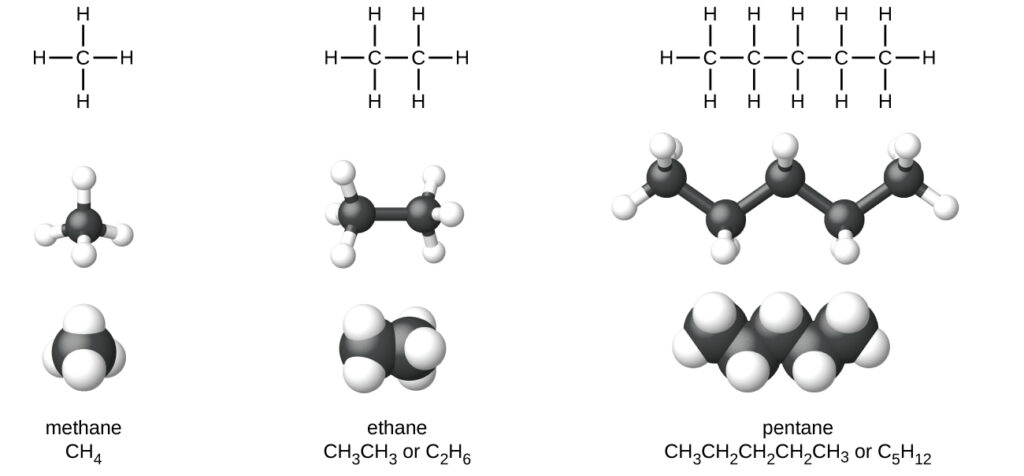
The smallest alkane is methane: CH4. The methane molecule is three dimensional, with the H atoms in the positions of the four corners of a tetrahedron.
The next-largest alkane is ethane. It has two C atoms that are covalently bonded to each other.
The general formula for an alkane is CnH2n+2. This information is on your information sheet.
How to name the straight chain alkanes?
Simple, straight-chain alkanes are named STEM-SUFFIX as follows:
- Count the number of carbons in the chain.
- Use the table below (in your information sheet) to identify the stem which corresponds with the number of carbons.
- The suffix is “ -ane” for alkanes
What are the empirical formulae?
Empirical formulas are often determined by experimentation.
An empirical formula tells us the relative ratios of different atoms in a compound.
| Substance | Molecular Formula | Empirical Formula |
| Benzene | C6H6 | CH |
| Ethene (Acetylene) | C2H2 | CH |
| Glucose | C6H12O6 | CH2O |
| Water | H2O | H2O |
What are molecular formulae?
Molecular formulas give the kind and number of atoms of each element present in the molecular compound.
The molecular formula of methane is CH4 and that is also its empirical formula. Sometimes, however, the molecular formula is a simple whole number multiple of the empirical formula.
The molecular formula of acetic acid is C2H4O2. The molecular formula of glucose is C6H12O6. The structures of both molecules are shown.
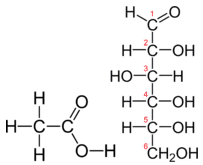
They are very different compounds, yet both have the same empirical formula of CH2O.
What are structural formulae?
The structural formula of a chemical compound is a graphic representation of the molecular structure.
The structural formulae that you draw will be Lewis structures.
Lewis structures are not intended to indicate the geometry of molecules. Notice that the carbon atoms in the structural models (the ball-and-stick and space-filling models) of the pentane molecule do not lie in a straight line.
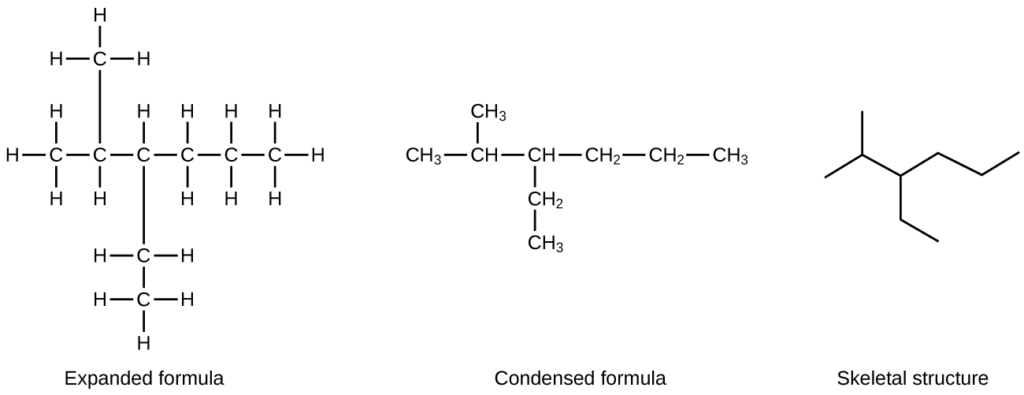
What are condensed structural formulae?
The structures of organic molecules may also be represented in a less detailed way by condensed structural formulas (or simply, condensed formulas).
A condensed formula is written to suggest the bonding in the molecule.
These formulas sometimes have the appearance of a Lewis structure from which most or all of the bond symbols have been removed.
Isomers
What are isomers?
Structural isomers are molecules that have the same molecular formula but different structures.
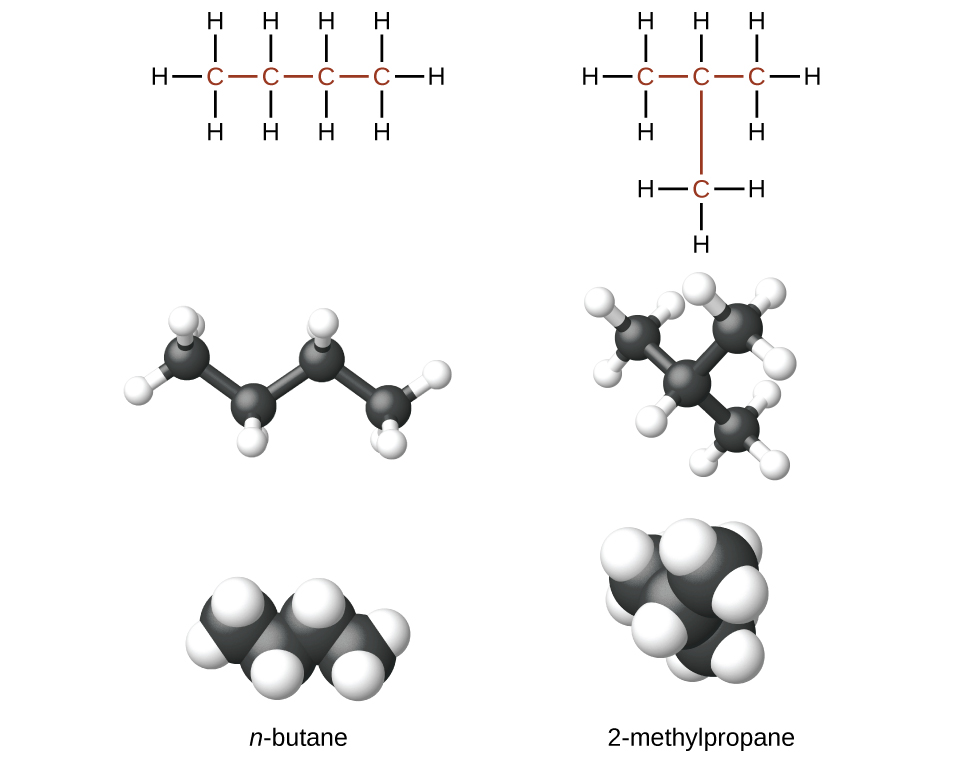
The two alkanes shown have the formula C4H10: They are isomers of butane called n-butane and 2-methylpropane (or isobutane)
How to spot isomers?
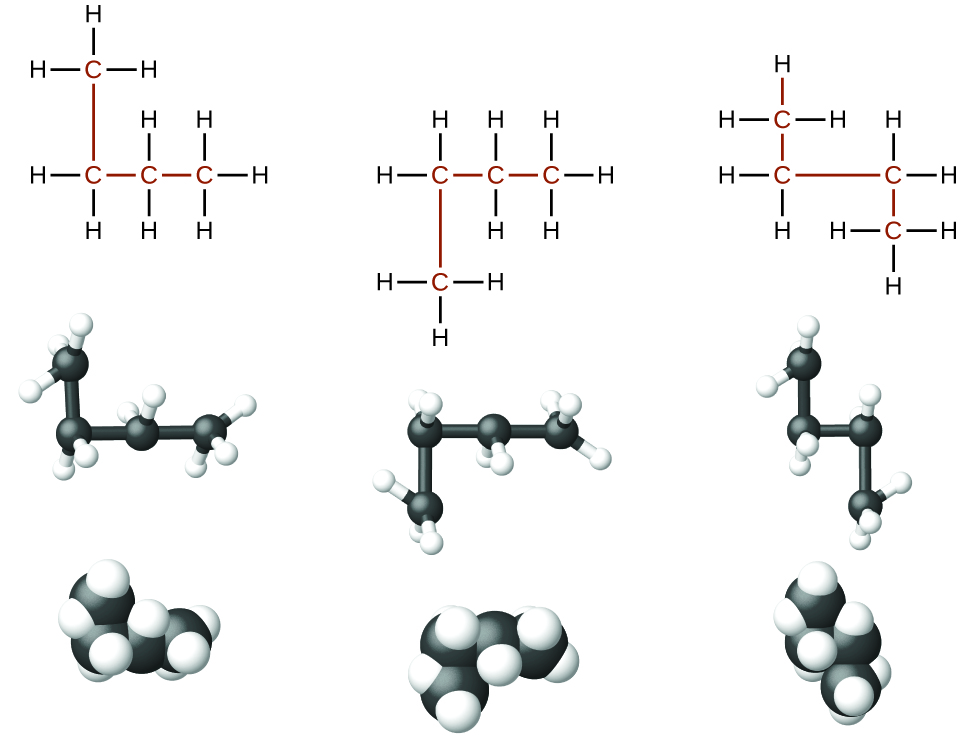
Rotation about the carbon-carbon bond can results in many different possible molecular conformations. For this reason, Identifying isomers from Lewis structures is not as easy as it looks.
Lewis structures that look different may actually represent the same isomers. For example, the three structures in all represent the same molecule, n-butane, and hence are not different isomers. They are identical because each contains an unbranched chain of four carbon atoms.
Alkane nomenclature
What is the IUPAC system?
The International Union of Pure and Applied Chemistry (IUPAC) has devised a system of nomenclature that begins with the names of the alkanes and can be adjusted from there to account for more complicated structures.
Teaching usually lags behind changes to official nomenclature by a few years, so marking will be quite flexible. For example, most students learn to identify the longest chain containing a double or triple bond, and name the compound according to that longest chain. This is no longer recommended by IUPAC.
The systemic name for all hydrocarbons essentially has three parts:
PREFIX-STEM-SUFFIX
How to name alkanes?
- Identify the longest chain of carbon atoms in its structure.
The stem is based on the number of carbon atoms in this chain.
The suffix is -ane for alkanes
E.g. a two-carbon chain is called ethane; a three-carbon chain, propane; etcetera.
How to name alkanes? II
- Determine which end is nearest to a branch, a double bond or a triple bond. (A double or triple bond takes precedence over a branch if they are equidistant from either end of the chain.)
- Number the carbon atoms from the end chosen
- Add prefixes to the name of the longest chain to indicate the positions and names of side chains and functional groups (substituent). The position of a substituent is identified by the number of the carbon atom it is bonded to in the chain.
The names of the side chains and functional groups can be found on your information sheet.
- Multiple substituents are named individually and placed in alphabetical order at the front of the name.
Because the carbon atom numbering begins at the end closest to a substituent, the longest chain of carbon atoms is numbered in such a way as to produce the lowest number for the substituents. - When two or more identical branches occur on different carbon atoms, the prefixes di-, tri- and tetra- are used.
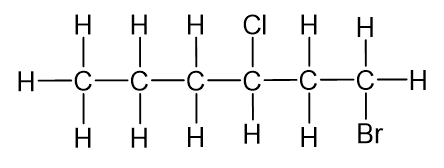
The prefixes di, tri, tetra etc., used to designate several groups of the same kind, are not considered when alphabetizing.
E.g. 2,3-dibromo-4-chloroheptane not 4-chloro-2,3-dibromoheptane
Avoid the following common errors when naming hydrocarbons:
- not identifying the longest chain possible
- not listing the side branches in alphabetical order
- omitting the prefixes di-, tri- and tetra- when they are required
What are the alkyl groups?
We call a substituent that contains one less hydrogen than the corresponding alkane an alkyl group. The name of an alkyl group is obtained by dropping the suffix -ane of the alkane name and adding -yl:
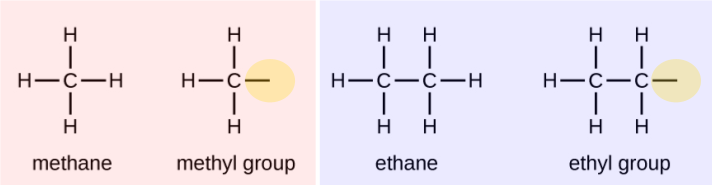
The open bonds in the methyl and ethyl groups indicate that these alkyl groups are bonded to another atom.
Alkyl halides
Why care about alkyl halides?
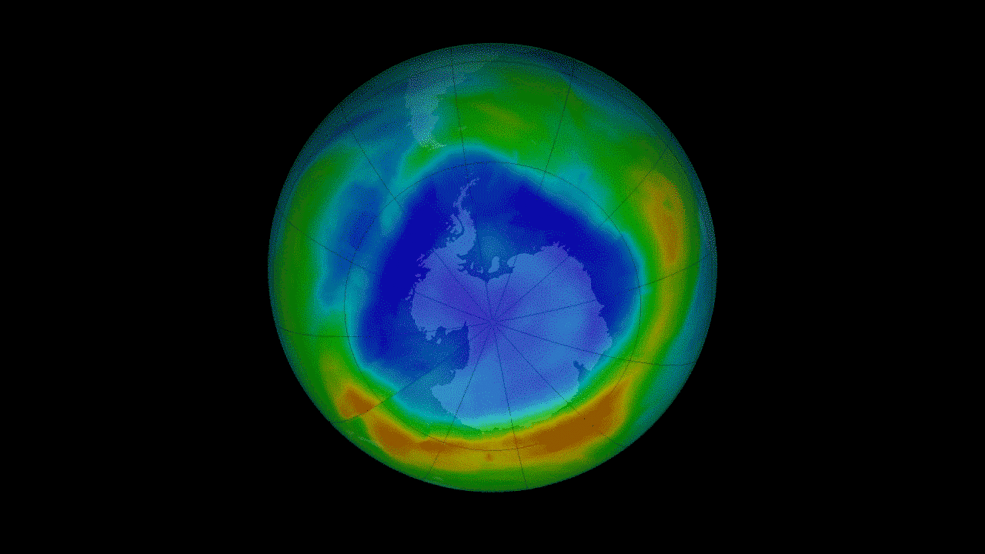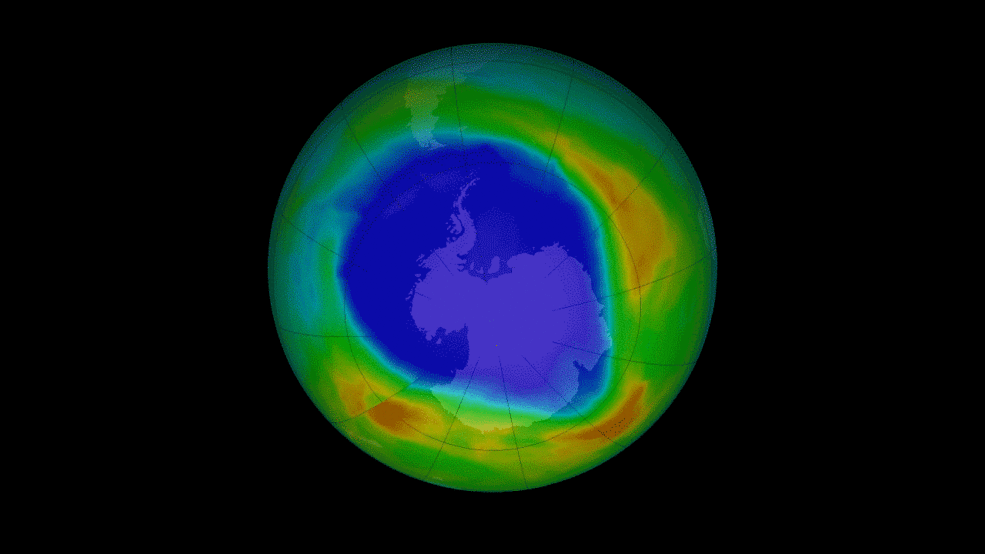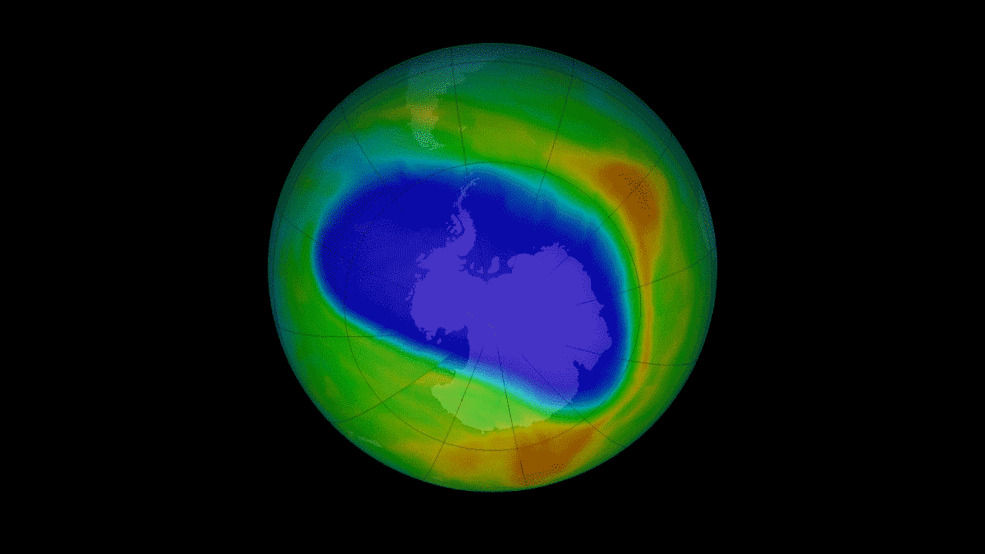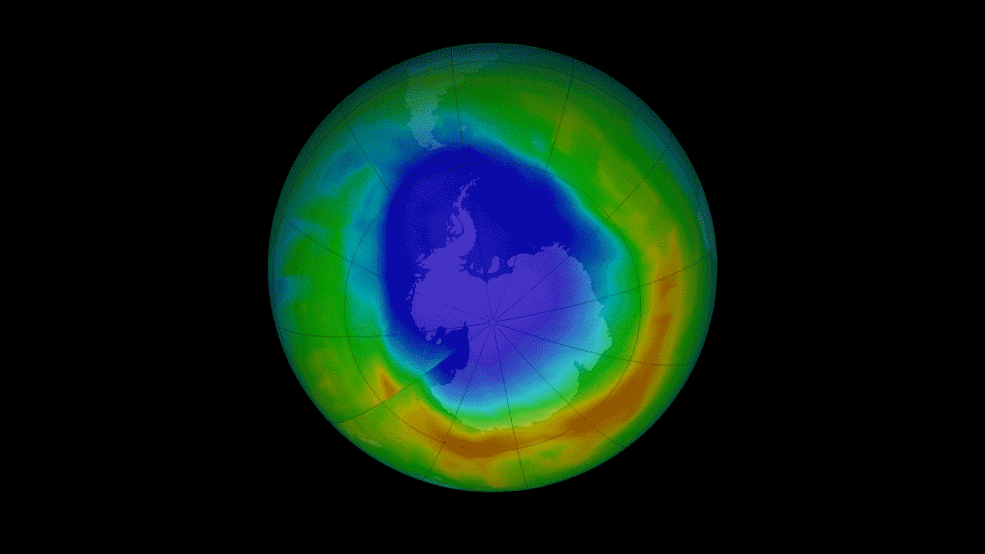
CFCs and newer refrigerants are alkyl halides, or alkene halides.
CFCs e.g. Trichlorofluoromethane (banned since ‘96) catalyse the destruction of ozone.
Some replacements have high greenhouse warming potential.
1,1,1,2-Tetrafluoroethane, known as R134a, is commonly used in heat pumps (air conditioners).
What are alkyl halides?
Alkyl halides are compounds in which one or more hydrogen atoms in an alkane have been replaced by halogen atoms (fluorine, chlorine, bromine or iodine).
They are named in a similar way to alkyl groups except they use a suffix ending in ‘o’ e.g. bromo, iodo, chloro. The alphabetical rule still applies.
How to draw structural formulae of alkanes?
- Draw the backbone of carbons, including any multiple bonds.
- Add dashes to indicate where attachments will go.
- Draw side chains and functional groups. (methyl, ethyl, Cl, Br etc.)
- Side chains can be drawn in full, or in a semi-structural format eg –C3H7.
- When drawing rough diagrams, a dash without other symbols or formulae attached indicates a hydrogen is attached.
It is important to remember in assignments and exams to write in every hydrogen.
Alkenes
What are saturated and unsaturated hydrocarbons?
Saturated hydrocarbons contain only single carbon-carbon bonds. Unsaturated hydrocarbons contain double or triple carbon-carbon bonds.
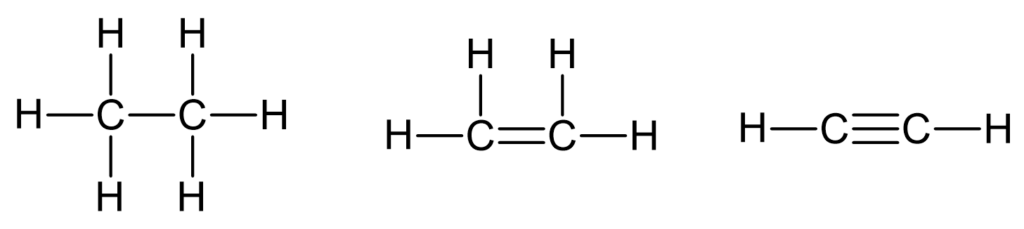
What are alkenes?
The alkenes make up a family of hydrocarbons that each contain one double bond between two carbon atoms. So they are unsaturated hydrocarbons.
Notice that each of these names ends with -ene. This is the characteristic ending used for the alkenes.
The alkenes have a general formula, CnH2n, and are another example of a homologous series.
Where is the double bond?
From butene onwards there are multiple places the double bond can appear within the chain.
As a result, we must name alkenes to indicate the position of the double bond using a number.
The number is placed between the stem and the suffix “-ene” using hyphens.
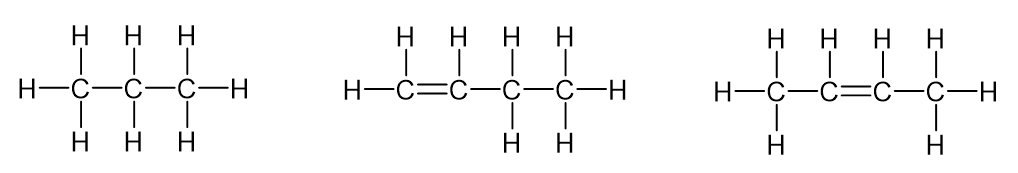
Why isn’t there a but-3-ene?
How to name alkenes? I
- Find the longest carbon chain in the molecule that contains both carbons of the double bond.
- The stem must be numbered so that the double bond number is the smallest possible. If the double bond is equidistant from both ends of the parent chain, number from the end which gives the substituents the lowest possible number. Note the number of the double bond takes precedence over any side groups.
- Place the location number of the double bond directly before the -ene suffix of alkene names. The location number indicates the position of the first carbon of the double bond.
- Add substituents and their position to the alkene as prefixes. Remember substituents are written in alphabetical order.
- Multiple double bonds have di, tri etc used and multiple location numbers are required,
- Then apply the same rules as with the alkanes.
Cyclic hydrocarbons
What are cycloalkanes?
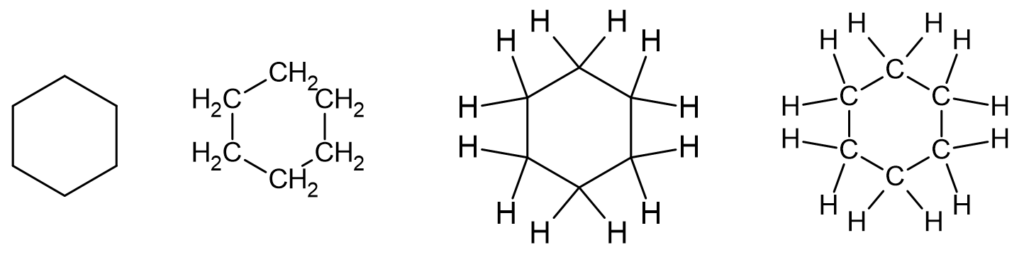
The carbon backbone of some hydrocarbons can form rings. Such compounds are known as cyclic hydrocarbons and have the prefix cyclo- before the stem.
Cycloalkanes have the general formula of CnH2n
Understanding cycloalkanes and their properties are crucial in that many of the biological processes that occur in most living things have cycloalkane-like structures.
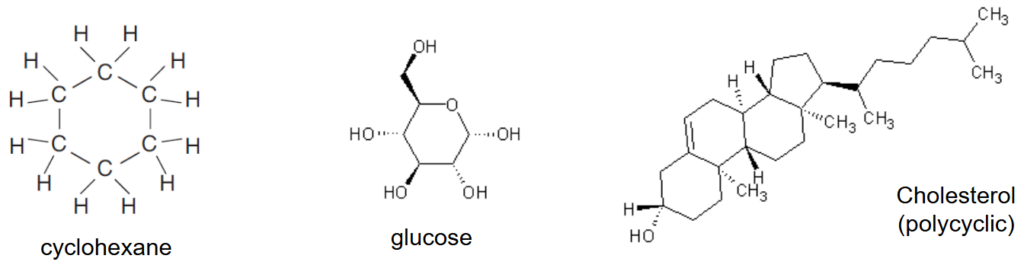
How are cycloalkanes drawn?
How to name cyclic hydrocarbons with side groups?
As with straight alkanes, these can have alkyl or functional groups on them and number starts on the carbon which makes the numbers of the alkyl or functional groups the lowest.
A carbon atom with multiple substituents should have a lower number than a carbon with only one substituent or functional group.
One way to make sure that the lowest number possible is assigned is to number the carbons so that when the numbers corresponding to the substituents are added, their sum is the lowest possible.
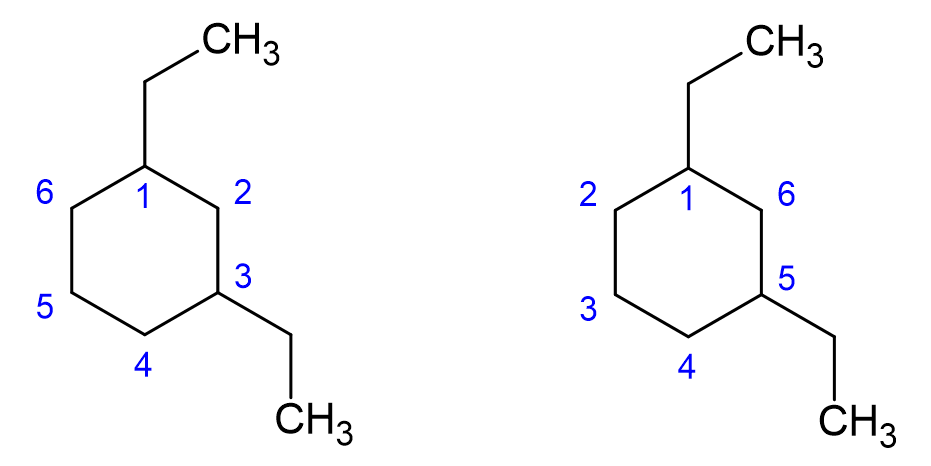
The stucture above is called 1-ethyl-3-methylcyclohexand, following the numbering scheme on the left (sum of numbers 1 + 3 = 4). The numbering scheme shown on the right results in 1-ethyl-5-methylcyclohexane (sum of numbers 1 + 5 = 6).
What are cycloalkenes?
Like alkanes, alkenes can also form rings. This means there are two less hydrogens as the end carbons bond with each other.
Thus, the general formula for a cyclic alkene is CnH2n-2
These are named with a ‘cyclo’ before the stem.
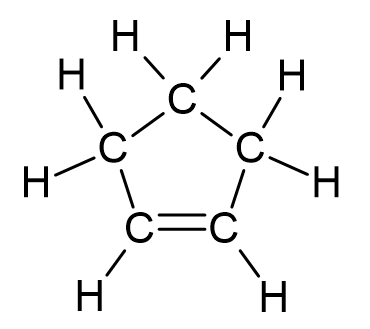
How to number the carbons in cycloalkenes?
Numbering begins on the carbon with the double bond and continues in the direction which will keep side group numbers the lowest.
How to name cycloalkenes? I
Because there are no chain ends in cycloalkenes, the double bond is assumed to be between C1 and C2 and its location number is not required in the name.
The direction of the numbering is determined by which will give the substituent closest to the double bond the lowest number.
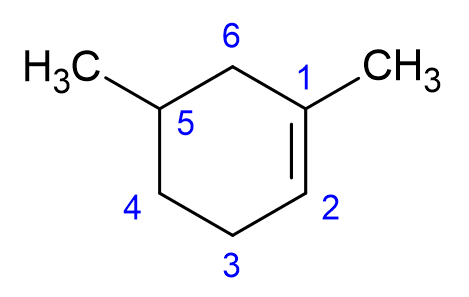
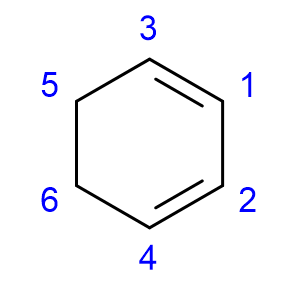
How to name cycloalkenes? advanced
If multiple double bonds are present, it may be necessary to include their location numbers in the name. One of the double bonds will be between C1 and C2 and the numbering direction is determined by which gives the remaining double bonds the lowest possible number.
Alkynes
What are alkynes?
Alkynes are members of a homologous series. The alkyne homologue has only carbon and hydrogen atoms in them but has the following characteristics:
There is at least one ㅡ C ☰ C ㅡ triple bond.
Thus, they are unsaturated hydrocarbons.
The general formula for an alkyne is CnH2n-2
Each member of the homologue differs from the previous by -CH2–
alkynes use the suffix –yne

How to name alkynes?
Find the longest carbon chain that includes both carbons of the triple bond.
Number the longest chain starting at the end closest to the triple bond.
The usual rules for naming and numbering side chains and functional groups apply.
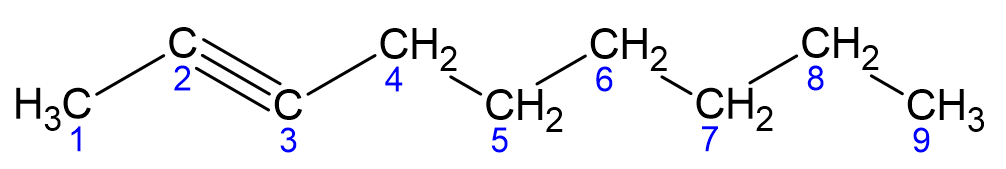
Finding isomers
How to find all the different isomers for a given molecular formula?
If you are given a molecular formula like C5H10, how to find the different isomers?
For alkanes, draw the structural formulae for these versions of the molecule:
- The simple unbranched version
- Versions with side branches
- Change the position of functional groups like chloro- or bromo- groups.
For alkenes and alkynes, draw the structural formulae for these versions of the molecule:
- The versions with double bonds in different positions
- Cyclic versions
- Are there versions with side branches?
- Can you change the position of functional groups like chloro or bromo groups?
How to use the general formulas to help?
Remember, the general formula of an alkene (CnH2n) is the same as the general formula of a cyclic alkane (CnH2n)!
Remember, the general formula of an alkyne (CnH2n-2) is the same as the general formula of a cyclic alkene (CnH2n-2)!
So if you are given a hydrocarbon that matches the general formula, you can easily find isomers!
Reactions of carbon compounds
What is combustion in alkanes, alkenes and alkynes?
Alkanes are relatively stable molecules, but heat or light will activate reactions that involve the breaking of C–H or C–C single bonds. Combustion is one such reaction:
Alkanes (as well as alkenes and alkynes) burn in the presence of oxygen, a highly exothermic reaction.
As a consequence, alkanes are excellent fuels. For example, methane, CH4, is the principal component of natural gas. Butane, C4H10, used in camping stoves and lighters is an alkane. Gasoline is a liquid mixture of continuous- and branched-chain alkanes, each containing from five to nine carbon atoms, plus various additives to improve its performance as a fuel. Kerosene, diesel oil, and fuel oil are primarily mixtures of alkanes with higher molecular masses.
What are the products of complete combustion?
When there is a plentiful supply of oxygen, the hydrocarbon will undergo complete combustion.
Complete combustion means that the only products are carbon dioxide and water – both in gas form.
The video shows an important test for the presence of carbon dioxide gas.
What is incomplete combustion?
When there is a limited supply of oxygen, the hydrocarbon will undergo incomplete combustion. The products will include:
- water (in the form of steam),
- possibly a combination of carbon (a solid called soot) AND/OR carbon monoxide and possibly some carbon dioxide.
Because the products can vary when combustion is incomplete, there are usually many possible answers to questions relating to the products formed when combustion is incomplete. For example:
C3H8(g) + 3O2(g) → 2CO(g)+ C(s) + 4H2O(g)
C3H8(g)+ 8 O2(g) → CO2(g) + 2CO(g) + C(s) + 4H2O(g)
How to write an equation showing incomplete combustion?
You might be asked to write an equation showing incomplete combustion of a hydrocarbon.
Remember, the reaction always produces water. I recommend trying to write an equation in which the ONLY other product is carbon (soot). Write the products first, then balance the reactants and products.
E.g. Write an equation for the incomplete combustion of C3H8.
Step 1: C3H8 + O2 → H2O + C
Step 2: C3H8(g) + 2O2(g) → 4H2O(g) + 3C(s)
Practice:
Write an equation for the incomplete combustion of ethane gas: C2H6 .
Write an equation for the incomplete combustion of butane gas: C4H10.
How to predict when incomplete combustion will happen?
Unsaturated hydrocarbons are more likely to undergo incomplete combustion than saturated hydrocarbons.
For saturated hydrocarbons, insufficient oxygen in the reaction mix results in incomplete combustion.
Generally, more O2 availability favours more CO2 as a product.
Questions will give you the clues to the product(s) formed.
| Oxygen (O2) availability | Combustion products |
| Very limited | Carbon (C) and water (H2O) |
| Limited | Carbon monoxide (CO) and water (H2O) |
| Plentiful (referred to as complete combustion) | Carbon dioxide (CO2) and water (H2O) |
Why do unsaturated hydrocarbons burn with a sooty flame?
The typically sooty flame produced when unsaturated hydrocarbons burn is commonly explained in this way:
“Unsaturated hydrocarbons (with double or triple bonds between carbon atoms) require more oxygen to undergo complete combustion”.
But why is this? You might read answers like ‘this is because the percentage of carbon in the saturated hydrocarbons is comparatively low’ or ‘in unsaturated compounds the carbon to hydrogen ratio is high and needs more oxygen’
Is this true? Let’s compare the combustion of ethane to the combustion of ethyne (acetylene):
C2H2 + 2.5O2 → 2CO2 + H2O
C2H6 + 3.5O2 → 2CO2 + 3H2O
The combustion reaction equations show that less, rather than more, oxygen is needed to combust an unsaturated compound.
You are not expected to be able to explain why unsaturated hydrocarbons require more oxygen to undergo complete combustion, explained below.
Why do unsaturated hydrocarbons produce soot? (advanced)
you can think of complete combustion versus incomplete combustion as the outcome of a race between soot formation and the formation of CO2, rather than being due to an overall deficiency of oxygen.
The presence of acetylene in a flame dramatically favours the formation of soot, by providing a competing reaction pathway to the traditional oxidation route to CO2.
The closer the starting ingredient’s structure is to that of acetylene (for example if it contains double or triple bonds) the more dominant that reaction will be, and therefore the more soot will be produced.
Source: https://edu.rsc.org/feature/clearing-the-air-around-smoke-formation/2500478.article
Substitution reactions
What are substitution reactions?
These are typical reaction of alkanes.
One or more of the alkane’s hydrogen atoms is replaced with a different atom or group of atoms.
No carbon-carbon bonds are broken in these reactions.
For example, the reaction between ethane and molecular chlorine depicted here is a substitution reaction:
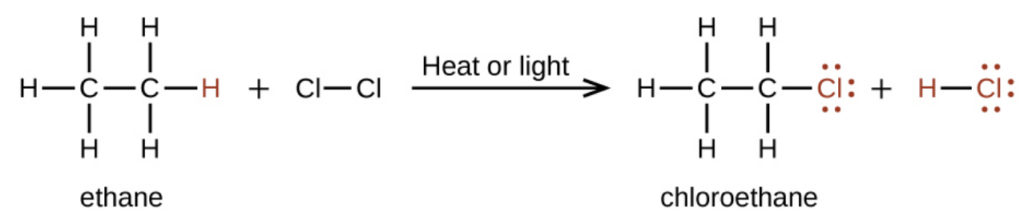
What are the products of halogenation reactions?
Substitution reactions happen in which hydrogen atoms in the alkane are replaced one at a time by chlorine atoms. You end up with a mixture of products.
For example, in the halogenation of methane, products can include chloromethane, dichloromethane, trichloromethane and tetrachloromethane.
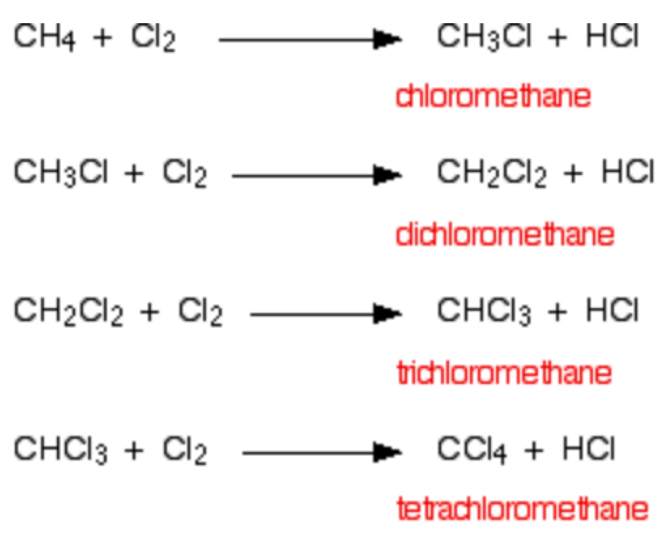
You need to be able to identify the first step in halogenation.
What is required for substitution reactions?
The halogenation of alkanes can be initiated with heat or light of a certain wavelength (especially UV).
In the reaction shown, soot is produced in one of the many complex side reactions.
Unless initiated by UV light, substitution reactions are usually VERY SLOW and thus initial observations may reveal no changes have occurred at all!
(Note that the reaction in the video is very fast because the flash light was very powerful)
Addition reactions
What are addition reactions?
Alkenes are much more reactive than alkanes because the C=C double bond is a reactive functional group.
Alkenes undergo a characteristic reaction in which the double bond is broken and replaced by two single bonds. This reaction is called an addition reaction.
For example, halogens add to the double bond in an alkene instead of replacing hydrogen, as occurs in an alkane:
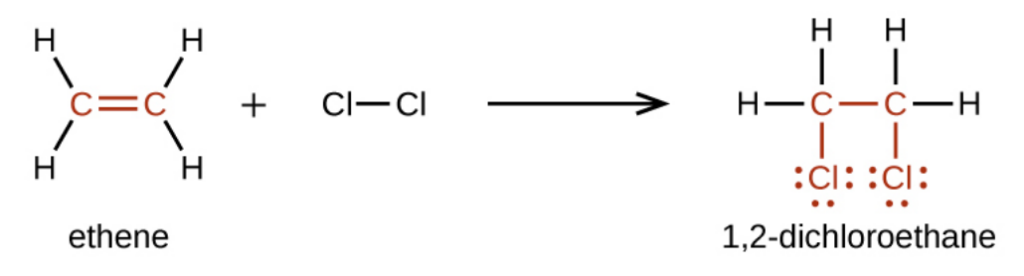
How do substitution and addition reactions compare?
| Type of hydrocarbon | Substitution reactions | Addition reactions |
| Alkanes | Slow, unless initiated by UV light | Impossible |
| Alkenes | Not typical | Rapid |
What compounds can react in addition reactions?
Alkenes react RAPIDLY with:
- Halogens (Cl2, Br2 or F2) (known as halogenation)
- Hydrogen (known as hydrogenation)
- Hydrogen halides (HCl, HBr, HF) (also an example of halogenation)
- Water (known as hydration)
The addition of hydrogen halides:
The addition reaction of a hydrogen halide to an alkene produces an alkyl halide as a product. For example:
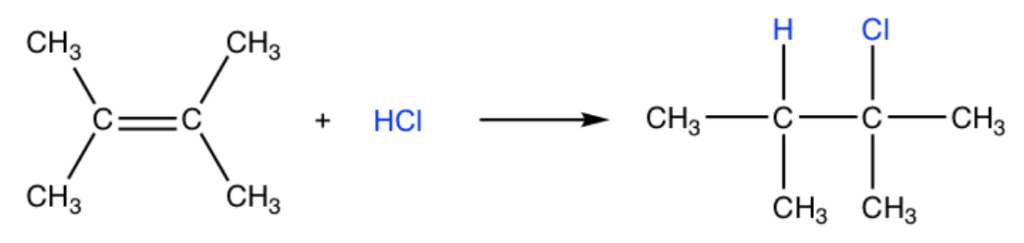
The addition of hydrogen
When alkenes react with hydrogen gas in the presence of a variety of metal catalysts, a hydrogen molecule will be added to the double bond in a way that each carbon atom bonds with one hydrogen atom. Such an addition reaction is called hydrogenation.
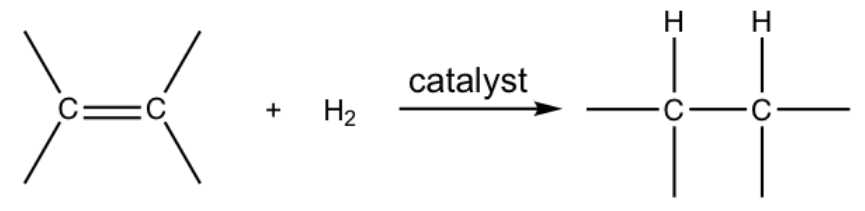
Bromine test for saturation
How to test for saturation?
A key feature when investigating the properties of hydrocarbons is to determine whether the compound is saturated or unsaturated. As burning organic chemical is often dangerous then Substitution/Addition testing is the best method to distinguish saturated and unsaturated organic chemical.
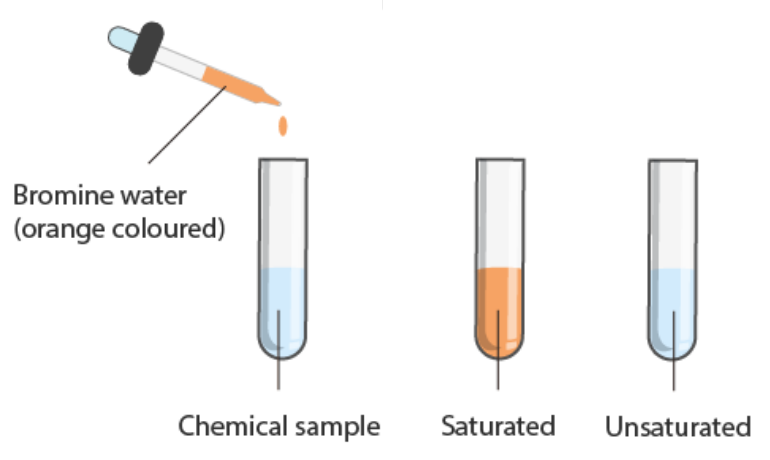
The simplest test is carried out by adding bromine (Br2) to the organic chemical.
Bromine in the elemental (un-combined) form is brown Br2.
When mixed with an unsaturated hydrocarbon (alkene or alkyne or cycloalkene) there is a very rapid ADDITION reaction and a colour change (brown to clear) is observed.
When mixed with a saturated hydrocarbon, no immediate reaction is observed.
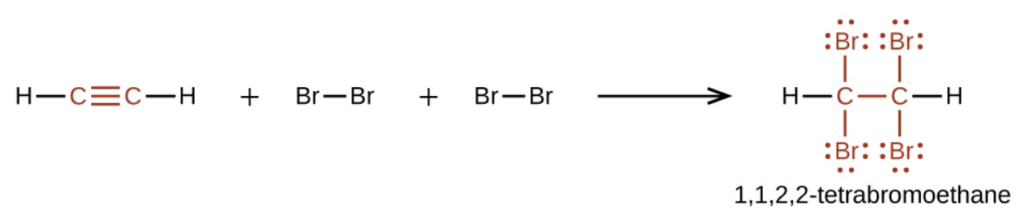
Why does the bromine test for saturation work?
The new compound formed in the addition reaction will be a “dibromo” or “tetrabromo” compound and these are colourless!
Although saturated compounds do eventually undergo a substitution reaction with bromine, the reaction is so slow that no significant colour change may be seen for many hours unless the combination is heated or exposed to UV light.
What are the gas tests?
Gas tests are in Criterion 7 but match the C8 concepts and reactions better.
- “Pop” test for hydrogen.
- Place a burning splint near the mouth of a test tube.
A “pop” will be heard if H2 was present
- Place a burning splint near the mouth of a test tube.
- Limewater test for CO2
- Bubble the gas through limewater
The limewater will turn milky if CO2 was present
- Bubble the gas through limewater
- Glowing splint test for O2
- Light a splint and let it go out (but it is still glowing)
Place the glowing splint into the test tube.
If the ember glows more brightly then O2 was present
- Light a splint and let it go out (but it is still glowing)
- Burning splint test
- Place a burning splint into the test tube. If the flame is extinguished, the gas:
- is not O2
- could be CO2
- Place a burning splint into the test tube. If the flame is extinguished, the gas: